|
(b) convert J of one photon to kJ/mol of photons: I will use 1 eV = 1.6022 x 10¯ 19 J for this problem. To solve this problem, you must know the relationship between electron-volts (eV) and Joules. For the 1.17 MeV photon, determine (a) the energy in Joules of one photon as well as (b) the energy produced in units of kJ per mole. This value is usually reported in kJ per mole: 9.98 x 10 5 kJ mol¯ 1Įxample #18: When it decays by beta decay, cobalt-60 also produces two gamma rays with energies of 1.17 MeV and 1.33 MeV. To get energy per mole, multiply the above value by Avogadro's Number: There was no need to take the wavelength to meters. I did this because the 1.2 Ångstrom value for the wavelength converts very easily into cm. Note also that I used 3.00 x 10 10 cm s¯ 1 for the speed of light. 
Calculate the energy of one mole of photons with this wavelength.ġ.2 Å x (10¯ 8 cm / 1 Å) = 1.2 x 10¯ 8 cm It's just a stylistic thing to write it the first way.Įxample #17: A particular x-ray has a wavelength of 1.2 Å. nm.īy the way, Eλ = hc can also be written as E = hc/λ. 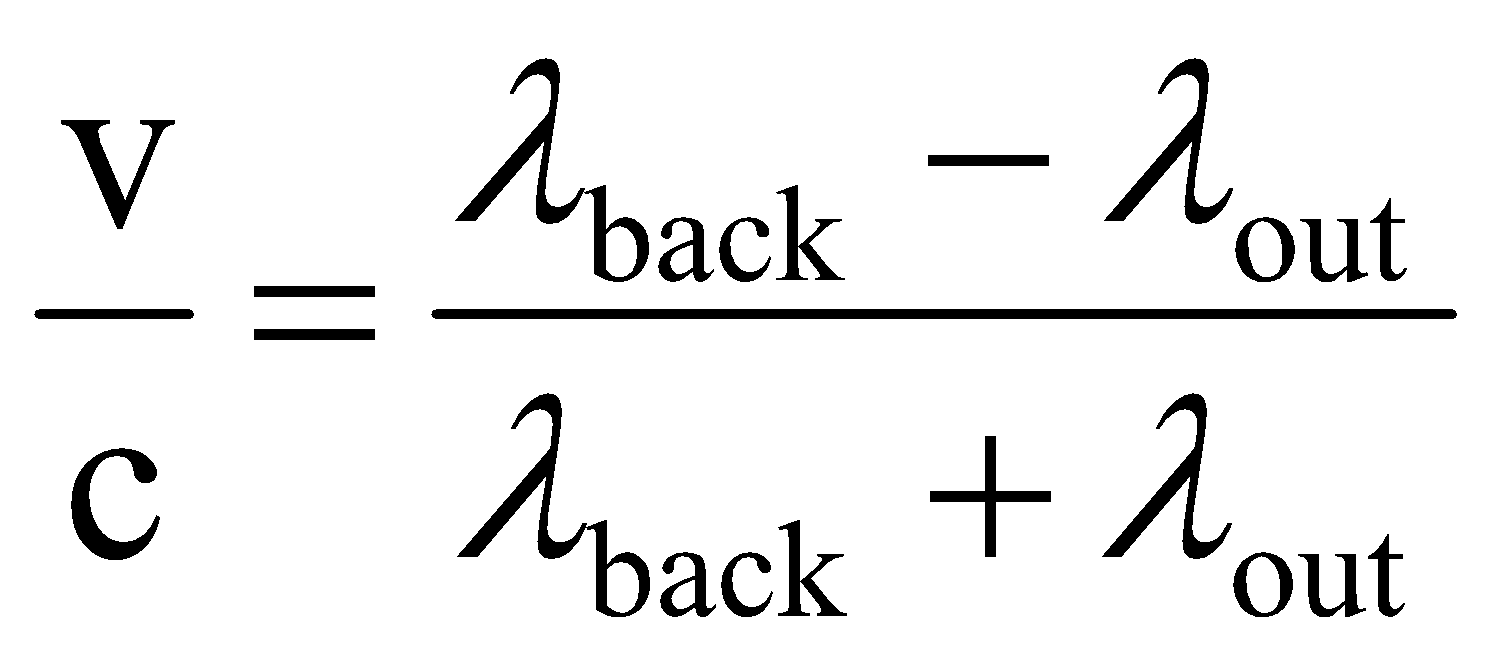
(3.82 x 10 -19 J) (λ) = (6.626 x 10 -34 J s) (3.00 x 10 8 m/s)Ĥ) You may do the math set up for converting from meters to nanometers. We will assume the energy given is for one photon.ģ.82 x 10 -22 kJ times (1000 J / 1 kJ) = 3.82 x 10 -19 JĢ) What you now do is combine these two equations: Problem #16: The energy of a particular color of green light is 3.82 x 10 -22 kJ. 
There are many who disagree that indigo deserves to be a color in the spectrum, so choosing blue as the answer seems the best way to go. Note that this chart is divided up into seven colors (the well-known Roy G. This is 462.3 nm (You may do the conversion to check it.)ģ) To determine the color, we look at a chart of wavelengths and colors. What color is the cesium flame?ġ) We combine E = hν and λν = c to arrive at: Problem #15: When a cesium salt solution is ionized in a Bunsen or Meeker burner, photons of energy 4.30 x 10 -19 J are emitted. (1 W = 1 J/s)Ģ) Determine energy of one photon at this wavelength:Į = / 5.32 x 10¯ 7 mģ) Determine power output in terms of photons:Ĥ J s¯ 1 / 3.736466 x 10¯ 19 J per photonĤ) Determine decimal percent pinhole is of total area of beam:ĥ) Determine photons through pinhole in one second: If the laser is pointed toward a pinhole with a diameter of 1.2 mm, how many photons will travel through the pinhole per second? Assume that the light intensity is equally distributed throughout the entire cross-sectional area of the beam. The diameter of the laser beam is 6.2 mm. Problem #14: An argon ion laser puts out 4.0 W of continuous power at a wavelength of 532 nm. 
What is the wavelength of light (in nm) that would be required to break the bond of one Cl 2 molecule. Problem #13: It requires 325 kJ to break one mole of Cl 2 bonds. In that case, multiply by Avogadro's Number, the divide by 1000 to arrive at the answer. Often, this type of question will ask for the energy in kJ/mol. Use Eλ = hc (with the speed of light as 3.00 x 10 8 m s¯ 1Į = / 6.62 x 10¯ 7 m Problem #12: What is the energy of light with a wavelength of 662 nm? What is the frequency in Hz of this gamma ray? A gamma ray emitted by an atom of Thallium-201 has an energy of 0.1670 million electron-volts. Problem #11: The radioactive isotope Thallium-201 is used in medical diagnosis and treatment. Problems #11 - 20 Wavelength-Frequency-Energy Problems #1 - 10 Go to Part Two of Light Equations Return to Part One of Light Equations Return to Electrons in Atoms menu Calculations between wavelength, frequency and energy - Probs 11-20Ĭalculations between wavelength, frequency and energy
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
